COMPARATIVE STUDIES OF THE CHEMICAL INTERACTION OF GUANIDINE WITH DIALDEHYDE CELLU-LOSE AND PECTIN
UDC 541.6.69:615.01
Abstract
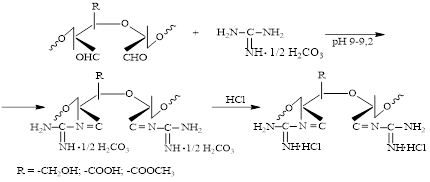
The presence of reactive aldehyde groups in the elementary units of dialdehyde polysaccharides enables to enter easily into a condensation reaction with reagents containing primary amino groups in the structure. This naturally expands the possibilities of obtaining new derivatives of polysaccharides with physiologically active properties. In this study, we synthesized azomethine derivatives of cellulose and pectin differing in the degree of substitution and the content of the nucleophilic reagent in the composition of the reaction products through chemical interaction of guanidine with macromolecules of dialdehyde polysaccharides. Therefore, regularities of the reaction of nucleophilic substitution of aldehyde groups of oxidized polysaccharides with guanidine were revealed. It was found that the interaction of guanidine amino groups with oxidized pectin in contrast to dialdehyde cellulose occurs through the formation of azomethine and ionic bonds. Moreover, the possibility of obtaining guanidine-containing derivatives of cellulose and pectin with different structural characteristics by varying the molar ratio of the nucleophile and the oxidation state of the starting polysaccharides was proved. The composition and structure of the reaction products were studied by physicochemical methods of analysis. The stability and rate of cleavage of guanidine groups from the macromolecules of dialdehyde cellulose and dialdehyde pectin were studied by hydrolysis of the synthesized samples in acidic and alkaline media.
Downloads
Metrics
References
Cumpstey I. ISRN Organic Chemistry, 2013, pp. 1–27. DOI: 10.1155/2013/417672.
Luo Y., Wang Q. International Journal of Biological Macromolecules, 2014, vol. 64, pp. 353–367. DOI: 10.1016/j.ijbiomac.2013.12.017.
Filatova A.V., Azimova L.B., Turayev A.S. Khimiya rastitel'nogo syr'ya, 2020, no. 1, pp. 33–39. DOI: 10.14258/jcprm.2020015485. (in Russ.).
Zhang L., Wang R., Liu R., Du X., Meng R., Liu L., Yao J. Cellulose, 2018, vol. 25, pp. 6947–6961. DOI: 10.1007/s10570-018-2083-x.
Almasi T., Jabbari K., Gholipour N., Mokhtari Kheirabadi A., Beiki D., Shahrokhi P., Akhlaghi M. International Journal of Biological Macromolecules, 2019, vol. 125, pp. 915–921. DOI: 10.1016/j.ijbiomac.2018.12.1.
Shomurotov Sh.A., Akhmedov O.R., Turayev A.S., Mamadullayev G.Kh. Khimiko-farmatsevticheskiy zhurnal, 2021, vol. 55, no. 6, pp. 67–71. DOI: 10.30906/0023-1134-2021-55-6-23-27. (in Russ.).
Ding W., Zhao P., Li R. Carbohydrate Polymers, 2011, vol. 83(2), pp. 802–807. DOI: 10.1016/j.carbpol.2010.08.057.
Sirvio J.A., Anttila A.K., Pirttila A.M., Liimatainen H., Kilpelainen I., Niinimaki J., Hormi O. Cellulose, 2014, vol. 21(5), pp. 3573–3583. DOI: 10.1007/s10570-014-0351-y.
Syutkin V.N., Nikolayev A.G., Sazhin S.A., Popov V.M., Zamoryanskiy A.A. Khimiya rastitel'nogo syr'ya, 2000, no. 1, pp. 5–25. (in Russ.).
Iozep A.A. Razrabotka putey modifikatsii prirodnykh polisakharidov s tsel'yu sozdaniya novykh biologicheski aktivnykh veshchestv: avtoref. dis. … dokt. farm. nauk. [Development of ways to modify natural polysaccharides in or-der to create new biologically active substances: abstract dis. … doc. farm. Sciences]. St. Petersburg, 1999, 48 p. (in Russ.).
Sarymsakov A.A., Nadzhimutdinov Sh., Tashpulatov Yu.T. Khimiya prirodnykh soyedineniy, 1998, no. 2, pp. 212–217. (in Russ.).
Akhmedov O.R., Shomurotov Sh.A., Turayev A.S. Khimiya rastitel'nogo syr'ya, 2021, no. 3, pp. 73–82. DOI: 10.14258/jcprm.2021038705. (in Russ.).
Akhmedov O.R., Sokhibnazarova Kh.A., Shomurotov Sh.A. Khimiya rastitel'nogo syr'ya, 2017, no. 3, pp. 227–331. DOI: 10.14258/jcprm.2017031729. (in Russ.).
Sun L., Yang S., Qian X., An X. Cellulose, 2020, vol. 27, pp. 8799–8812. DOI: 10.1007/s10570-020-03374-5.
Bychkovskiy P.M., Yurkshtovich T.L., Golub N.V., Solomevich S.O., Yurkshtovich N.K., Adamchik D.A. Vysoko-molekulyarnyye soyedineniya, 2019, vol. 61, no. 4, pp. 261–271. DOI: 10.1134/S2308113919040028. (in Russ.).
Syutkin V.N., Nikolayev A.G., Sazhin S.A., Popov V.M., Zamoryanskiy A.A. Khimiya rastitel'nogo syr'ya, 1999, no. 2, pp. 91–102. (in Russ.).
Tunik T.V., Nemchenko U.M., Ganenko T.V., Yurinova G.V., Dzhioyev Yu.P., Sukhov B.G., Zlobin V.I., Trofimov B.A. Izvestiya RAN. Seriya fizicheskaya, 2019, vol. 83(3), pp. 408–414. DOI: 10.1134/S0367676519030268. (in Russ.).
Guben-Veyl'. Metody organicheskoy khimii. Moscow, 1967, vol. 2, 1032 p. (in Russ.).
Anan'yeva Ye.P., Baranov S.S., Karavayeva A.V., Borisenko M.S., Solovskiy M.V., Zakharova N.V., Prazdniko-va T.A., Tarabukina Ye.B. Antibiotiki i khimioterapiya, 2014, vol. 59, no. 11–12, pp. 3–6. (in Russ.).
Borisenko M.S. Vodorastvorimyye reaktsionnosposobnyye sopolimery N-vinilpirrolidona i sopolimery akrilovoy kisloty s 2-oksietilmetakrilatom kak nositeli protivotuberkuloznykh preparatov: dis. … kand. khim. nauk. [Water-soluble reactive copolymers of N-vinylpyrrolidone and copolymers of acrylic acid with 2-hydroxyethyl methacrylate as carriers of anti-tuberculosis drugs: diss. … cand. chem. Sciences]. St. Petersburg, 2019, 153 p. (in Russ.).
Solovskiy M.V., Nikol'skaya N.V., Zaikina N.A. Khimiko-farmatsevticheskiy zhurnal, 2002, vol. 36, no. 2, pp. 9–13. (in Russ.).
Copyright (c) 2022 chemistry of plant raw material

This work is licensed under a Creative Commons Attribution 4.0 International License.

This work is licensed under a Creative Commons Attribution 4.0 International License.
The authors, which are published in this journal, agree to the following conditions:
1. Authors retain the copyright to the work and transfer to the journal the right of the first publication along with the work, at the same time licensing it under the terms of the Creative Commons Attribution License, which allows others to distribute this work with the obligatory indication of the authorship of this work and a link to the original publication in this journal .
2. The authors retain the right to enter into separate, additional contractual agreements for the non-exclusive distribution of the version of the work published by this journal (for example, to place it in the university depository or to publish it in a book), with reference to the original publication in this journal.
3. Authors are allowed to post their work on the Internet (for example, in a university repository or on their personal website) before and during the review process of this journal, as this may lead to a productive discussion, as well as more links to this published work.











