THERMODYNAMIC ANALYSIS OF BIOCHAR GASIFICATION
UDC 662.765:536.7
Abstract
In this research, a thermodynamic analysis of the gasification process of biochar obtained by pyrolysis of wood biomass was carried out. Thermodynamic functions, such as enthalpies and entropies of the formation of biochar, water vapor, carbon oxide, and hydrogen at various temperatures and pressures were determined. The thermodynamic (TD) studies showed that the gasification process is endothermic, while the entropy of this process is positive. From TD calculations, it follows that the gasification process of biochar cannot be carried out at normal pressure if the temperature of this process is below boundary Tb=895 K because the thermodynamic Gibbs potential under these conditions has a positive value. Thus, the gasification process of the biochar can be implemented only at quite high temperatures. It has been established that increasing the gasification temperature above Tb increases the equilibrium constant, which should enhance the yield of biogas. On the other hand, pressure is a negatory factor for the gasification process, since its increase leads to a decrease in the equilibrium constant. However, the negative impact of pressure can be compensated by the rise in gasification temperature.
Downloads
References
Ang T-Z., Salem M., Kamarol M. et al. Energy Strategy Reviews, 2022, vol. 43, 100939. https://doi.org/10.1016/j.esr.2022.100939.
Lee S., Speight J.G., Loyalka S.K. Handbook of Alternative Fuel Technologies. 1-st ed. CRC Press: Boca Raton, 2007, 568 p.
Energy consumption from fossil fuels and renewables in the United States from 2000 to 2023. URL: https://www.statista.com/statistics/184024/us-energy-consumption-from-fossil-fuels-and-renewables-since-1999/.
Energy statistics – an overview. Eurostat, 2024. URL: https://ec.europa.eu/eurostat/statistics-explained/index.php?title=Energy_statistics_-_an_overview#:~:text=Renewable%20energies%20accounted%20for%20the,renewable%20waste%20(2.4%20%25).
Rashmi R., Tripathi T., Pandey S., Kumar S. IJRASET, 2024, vol. 12, pp. 1079–1089. https://doi.org/10.22214/ijraset.2024.64808.
Ioelovich M. Sci. Environ., 2020, vol. 3, pp. 147–152.
Klemm D., Heublein B., Fink H.-P., Bohn A. Angew. Chem., 2005, vol. 44, pp. 2–37. https://doi.org/10.1002/anie.200460587
Ioelovich M. World J. Adv. Res. Reviews, 2024, vol. 24, pp. 1295–1338. https://doi.org/10.30574/wjarr.2024.24.1.3145.
Tursi A. Biofuel Res. Journal, 2019, vol. 22, pp. 962–979. https://doi.org/10.18331/BRJ2019.6.2.3.
Saidur R., Abdelaziz E.A., Demirbas A. et al. Renew. Sustain. Energy Reviews, 2011, vol. 15, pp. 2262–2289. https://doi.org/10.1016/j.rser.2011.02.015.
Ioelovich M. Bioresources, 2015, vol. 10, pp. 1879–1914. https://doi.org/10.15376/biores.10.1.1879-1914.
Damartzis T., Zabaniotou A. Renew. Sustain. Energy Reviews, 2011, vol. 15, pp. 366-378. https://doi.org/10.1016/j.rser.2010.08.003.
Song H., Yang G., Xue P. et al. Appl. Energy Combust. Sci., 2022, vol. 10, 100059. https://doi.org/10.1016/j.jaecs.2022.100059.
Alvarado-Flores J.J., Alcaraz-Vera J.V., Ávalos-Rodríguez M.L. et al. Energies, 2024, vol. 17, pp. 1–21.
Bounaceur A., Gautherot L.P., Tschamber V. et al. Solid Fuel Chemistry, 2020, vol. 54, pp. 239–250. https://doi.org/10.3103/S0361521920040035.
Al-Rahbi A.S., Williams P.T. Waste Disposal & Sustainable Energy, 2022, vol. 4, pp. 75–89. https://doi.org/10.1007/s42768-022-00103-5.
Schildhauer T.J., Biollaz S.M.A. Synthetic Natural Gas: From Coal, Dry Biomass, and Power-to-Gas Applications. John Wiley & Sons: Hoboken, 2016, 328 p.
Zhang J., Weng X., Han Y. et al. J. Energy Chemistry, 2013, vol. 22, pp. 459–467. https://doi.org/10.1016/S2095-4956(13)60060-1.
Koper G.J.M. An Introduction to Chemical Thermodynamics. 2nd ed. VSSD: Leiden, 2008, 212 p.
Klotz I.M., Rosenberg R.M. Chemical Thermodynamics. Basic Concepts and Methods. 7th ed. John Wiley & Sons Inc.: Hoboken, 2008, 588 p.
Sluiter J.B., Ruiz R.O., Scarlata C.J., Sluiter A.D., Templeton D.W. J Agric. Food Chem., 2010, vol. 58, pp. 9043–9053. https://doi.org/10.1021/jf1008023.
Eisermann W., Johnson P., Conger W.L. Fuel Proces. Technol., 1980, vol. 3, pp. 39–53. https://doi.org/10.1016/0378-3820(80)90022-3.
Żogała A. J. Sustain. Mining, 2014, vol. 13, pp. 30–38. https://doi.org/10.7424/jsm140205.
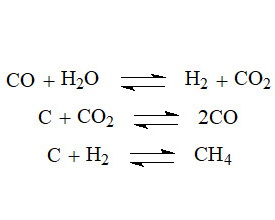
Copyright (c) 2026 Chemistry of plant raw material

This work is licensed under a Creative Commons Attribution 4.0 International License.

This work is licensed under a Creative Commons Attribution 4.0 International License.
The authors, which are published in this journal, agree to the following conditions:
1. Authors retain the copyright to the work and transfer to the journal the right of the first publication along with the work, at the same time licensing it under the terms of the Creative Commons Attribution License, which allows others to distribute this work with the obligatory indication of the authorship of this work and a link to the original publication in this journal .
2. The authors retain the right to enter into separate, additional contractual agreements for the non-exclusive distribution of the version of the work published by this journal (for example, to place it in the university depository or to publish it in a book), with reference to the original publication in this journal.
3. Authors are allowed to post their work on the Internet (for example, in a university repository or on their personal website) before and during the review process of this journal, as this may lead to a productive discussion, as well as more links to this published work.















