STUDY OF THE FEATURES OF REACTIONS FOR OBTAINING LOW-MOLECULAR CHITOSAN
UDC 547.99
Abstract
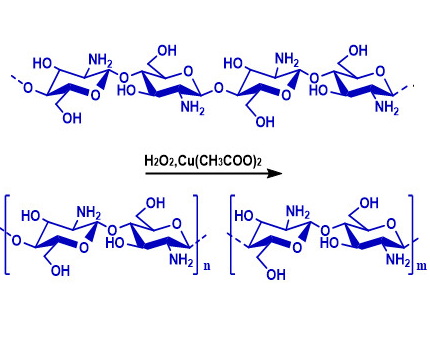
The reaction of radical depolymerization in the presence of hydrogen peroxide was studied with a chitosan sample with a degree of polymerization of 926, a molecular weight of 150 kDa, and a polydispersity index of 2.25. The reaction was carried out in the presence of Cu(CH3COO)2 (0.096 mg/ml), hydrogen peroxide solutions with different concentrations (9.0–1.0%), at a temperature of 60 °C for 10–60 min, and as a result of the reaction, low-molecular samples of chitosan and chitooligosaccharides with different molecular parameters and a yield of 6.4–83.4% were obtained. It was shown that as a result of the depolymerization reaction with hydrogen peroxide solutions with a low concentration, samples with low values of the degree of polymerization, polydispersity index and a high yield are obtained. It was found that in order to obtain low-molecular water-soluble chitosan samples, the reaction is advisable to carry out with a 3.0% hydrogen peroxide solution for 30–40 minutes. The structure of the obtained samples was studied by IR and 13C NMR spectroscopy. It was found that the IR spectra of chitosan and its depolymerization products are similar. Absorption in the regions of 1153, 1066 and 1031 cm-1 of the spectrum is due to the stretching vibrations of C-O-C groups, and absorption in the region of 898 cm-1 is characteristic of β-(1,4)-glycosidic bonds of chitosan. This indicates that during the radical depolymerization reaction no side reactions such as ring opening, dehydration occur, and the main structure of the polysaccharide chain is preserved. These results indicate that radical depolymerization of chitosan with the participation of hydrogen peroxide occurs due to the rupture of β-(1,4)-glycosidic bonds in the polysaccharide chain.
Downloads
References
Amonova D.M. i dr. Khimiya rastitel'nogo syr'ya, 2022, no. 2, pp. 51–60. https://doi.org/10.14258/jcprm.2022029551. (in Russ.).
Muhitdinov B., Heinze T., Turaev A., Koschella A., Normakhamatov N. European Polymer Journal, 2019, vol. 119, pp. 181–188. https://doi.org/10.1016/j.eurpolymj.2019.07.030.
Azimova L.B., Filatova A.V., Turayev A.S., Dzhurabayev D.T. Khimiya rastitel'nogo syr'ya, 2021, no. 3, pp. 115–122. https://doi.org/10.14258/jcprm.2021039173. (in Russ.).
Muxika A., Etxabide A., Uranga J., Guerrero P., de la Caba K. International journal of biological macromolecules, 2017, vol. 105, pp. 1358–1368. https://doi.org/10.1016/j.ijbiomac.2017.07.087.
Durante-Salmerón D.A., Fraile-Gutiérrez I., Gil-Gonzalo R., Acosta N., Aranaz I., Alcántara A.R. Catalysts, 2024, vol. 14, no. 6, 371. https://doi.org/10.3390/catal14060371.
El-Araby A., Janati W., Ullah R., Ercisli S., Errachidi F. Frontiers in Chemistry, 2024, vol. 11, 1327426. https://doi.org/10.3389/fchem.2023.1327426.
Kozma M., Acharya B., Bissessur R. Polymers, 2022, vol. 14, no. 19, 3989. https://doi.org/10.3390/polym14193989.
Abo Elsoud M.M., El Kady E.M. Bulletin of the National Research Centre, 2019, vol. 43, no. 1, pp. 1–12. https://doi.org/10.1186/s42269-019-0105-y.
Vanashi A.K., Ghasemzadeh H. International Journal of Biological Macromolecules, 2022, vol. 199, pp. 348–357. https://doi.org/10.1016/j.ijbiomac.2021.12.150.
Abd El-Hack M.E., El-Saadony M.T., Shafi M.E., Zabermawi N.M., Arif M., Batiha G.E, Khafaga A.F., Abd El-Hakim Y.M., Al-Sagheer A.A. International Journal of Biological Macromolecules, 2020, vol. 164, pp. 2726–2744. https://doi.org/10.1016/j.ijbiomac.2020.08.153.
Su J., Liu Ch., Sun A., Yan J., Sang F., Xin Y., Zhao Y., Wang Sh., Dang Q. International Journal of Biological Macromolecules, 2025, vol. 306 (2), 141570. https://doi.org/10.1016/j.ijbiomac.2025.141570.
Mawazi S.M., Kumar M., Ahmad N., Ge Y., Mahmood S. Polymers, 2024, vol. 16, no. 10, 1351. https://doi.org/10.3390/polym16101351.
Saini S., Dhiman A., Nanda S. Endocrine, Metabolic & Immune Disorders-Drug Targets (Formerly Current Drug Targets-Immune, Endocrine & Metabolic Disorders), 2020, vol. 20, no. 10, pp. 1611–1623. https://doi.org/10.2174/1871530320666200503054605.
Gonçalves C., Ferreira N., Lourenço L. Polymers, 2021, vol. 13, no. 15, 2466. https://doi.org/10.3390/polym13152466.
Niu Y., Hu W. Sustainable Environment Research, 2024, vol. 34, no. 1, 29. https://doi.org/10.1186/s42834-024-00236-8.
Tabassum N., Ahmed Sh., Ittisaf M.M., Rakid-Ul-Haque Md., Ali M.A. Cellulose, 2023, vol. 30, no. 14, pp. 8769–8787. https://doi.org/10.1007/s10570-023-05372-9.
Petit A.C., Noiret N., Sinquin C., Ratiskol J., Guézennec J., Colliec-Jouault S. Carbohydrate polymers, 2006, vol. 64, no. 4, pp. 597–602. https://doi.org/10.1016/j.carbpol.2005.11.016.
Ustyuzhanina N.E., Bilan M.I., Anisimova N.Yu., Dmitrenok A.S., Tsvetkova E.A., Kiselevskiy M.V., Ni-fantiev N.E., Usov A.I. Carbohydrate Polymers, 2022, vol. 281, 119072. https://doi.org/10.1016/j.carbpol.2021.119072.
Wang M., Veeraperumal S., Zhong S., Cheong K.L. Foods, 2023, vol. 12, no. 4, 878. https://doi.org/10.3390/foods12040878.
Liu Y., Harnden K.A., Van Stappen C., Dikanov S.A., Lu Y. Proceedings of the National Academy of Sciences, 2023, vol. 120, no. 43, e2308286120. https://doi.org/10.1073/pnas.2308286120.
Questell-Santiago Y.M., Galkin M.V., Barta K., Luterbacher J.S. Nature Reviews Chemistry, 2020, vol. 4, no. 6, pp. 311–330. https://doi.org/10.1038/s41570-020-0187-y.
Gu F., Liu H. Chinese Journal of Catalysis, 2020, vol. 41, no. 7, pp. 1073–1080. https://doi.org/10.1016/S1872-2067(20)63569-0.
Khademian E., Salehi E., Sanaeepur H., Galiano F., Figoli A. Science of the Total Environment, 2020, vol. 738, 139829. https://doi.org/10.1016/j.scitotenv.2020.139829.
Neji A.B. et al. Polymer Testing, 2020, vol. 84, 106380. https://doi.org/10.1016/j.polymertesting.2020.106380.
Valachová K., Šoltés L. International Journal of Molecular Sciences, 2021, vol. 22, no. 13, 7077. https://doi.org/10.3390/ijms22137077.
Hefni H.H.H., Saxena M., Mehta R., Bhojani G., Bhattacharya A. Polymer Bulletin, 2022, vol. 79, no. 6, pp. 4339–4360. https://doi.org/10.1007/s00289-021-03727-5.
Copyright (c) 2026 Chemistry of plant raw material

This work is licensed under a Creative Commons Attribution 4.0 International License.

This work is licensed under a Creative Commons Attribution 4.0 International License.
The authors, which are published in this journal, agree to the following conditions:
1. Authors retain the copyright to the work and transfer to the journal the right of the first publication along with the work, at the same time licensing it under the terms of the Creative Commons Attribution License, which allows others to distribute this work with the obligatory indication of the authorship of this work and a link to the original publication in this journal .
2. The authors retain the right to enter into separate, additional contractual agreements for the non-exclusive distribution of the version of the work published by this journal (for example, to place it in the university depository or to publish it in a book), with reference to the original publication in this journal.
3. Authors are allowed to post their work on the Internet (for example, in a university repository or on their personal website) before and during the review process of this journal, as this may lead to a productive discussion, as well as more links to this published work.















