SYNTHESIS AND STRUCTURAL CHARACTERISTICS OF TOSYL FUNCTIONALIZED GALACTOMANNANS
UDC 541.6.69.615.01
Аннотация
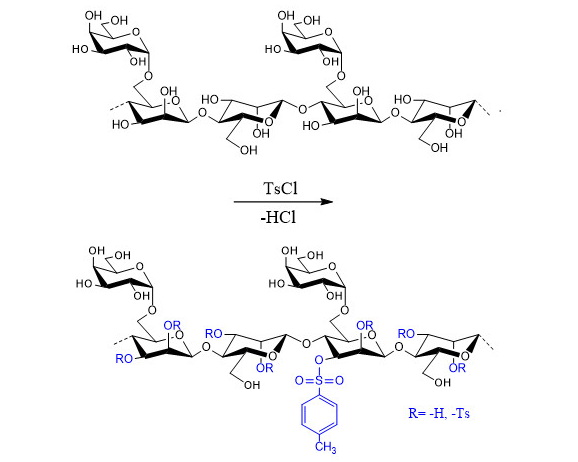
In this study, tosylation reactions of low-molecular-weight galactomannan samples (DP = 19, Mw = 9.1 kDa, Man/Gal = 1.79) were investigated in the presence of tosyl chloride. The tosylation reactions were carried out in different reaction media, including dimethyl sulfoxide (DMSO), N,N-dimethylformamide (DMF) and water environments. In the reactions performed in dimethyl sulfoxide and water media, no tosyl groups were detected in the products. However, when the reaction was conducted under the same conditions in N,N-dimethylformamide medium, galactomannan tosyl derivatives with degrees of substitution (DS) ranging from 0.37 to 0.71 were obtained. Tosylated galactomannan samples with a degree of substitution (DS) of 0.65 were obtained at 5 °C by using 3.0 mol of tosylating reagent per mol of GMU in N,N-dimethylformamide, achieving a yield of 77.1%. The structures of the low-molecular-weight galactomannan tosyl derivatives were investigated using UV, FTIR, and 13C NMR spectroscopy techniques. A characteristic absorption of the aromatic ring of the tosyl group was observed at 222 nm in the UV spectrum. FTIR spectroscopy confirmed the successful tosylation process through the characteristic -C=C- absorption bands of the tosyl aromatic ring in the range of 1590–1640 cm-1. Additionally, the symmetric and asymmetric stretching vibrations corresponding to the C-O-S and C-S bonds in the tosylated galactomannans were observed at 812 cm-1 and 667 cm-1, respectively. In the 13C NMR spectra of the obtained galactomannan tosyl derivatives, signals corresponding to the carbon atoms of the tosyl group were observed at 20.53; 125.42; 127.99; 129.51; and 142.54 ppm for TsC5, TsC3, TsC2, TsC4, and TsC1, respectively. Moreover, signals corresponding to the tosylated ManC2 and ManC3 carbon atoms were identified at 81.07 and 81.63 ppm. The spectroscopic analysis results indicated that the tosyl groups in the galactomannan tosyl derivatives were attached to the polysaccharide backbone via the ManC2 and ManC3 carbon atoms.
Скачивания
Литература
Li Z. W. et al. Polymers, 2022, vol. 14, no. 19, 4161. https://doi.org/10.3390/polym14194161.
Singh S. et al. Current Materials Science: Formerly: Recent Patents on Materials Science, 2023, vol. 16, no. 3, pp. 316–351. https://doi.org/10.2174/2666145416666221208150926.
Devadoss T. Journal of Molecular Structure, 2023, vol. 1289, 135850. https://doi.org/10.1016/j.molstruc.2023.135850.
Gericke M. et al. Carbohydrate Polymers, 2015, vol. 127, pp. 236–245. https://doi.org/10.1016/j.carbpol.2015.03.025.
Namnouad P. et al. New Journal of Chemistry, 2023, vol. 47, no. 30, pp. 14134–14141. https://doi.org/10.1039/D3NJ00830D.
Koschella A. et al. Molecules, 2020, vol. 25, no. 22, 5382. https://doi.org/10.3390/molecules25225382.
Potthast A. et al. Cellulose, 2002, vol. 9, pp. 41–53. https://doi.org/10.1023/A:1015811712657.
Zhang C. et al. The Journal of Physical Chemistry B, 2014, vol. 118, no. 31, pp. 9507–9514. https://doi.org/10.1021/jp506013c.
Ostlund A. et al. Biomacromolecules, 2009, vol. 10, no. 9, pp. 2401–2407. https://doi.org/10.1021/bm900667q.
Liebert T. American Chemical Society, 2010, pp. 3–54. https://doi.org/10.1021/bk-2010-1033.ch001.
Muhitdinov B. et al. Carbohydrate Polymers, 2017, vol. 173, pp. 631–637. https://doi.org/10.1016/j.carbpol.2017.06.033.
Muhitdinov B., Heinze T., Turaev A., Koschella A., Normakhamatov N. European Polymer Journal, 2019, vol. 119, pp. 181–188. https://doi.org/10.1016/j.eurpolymj.2019.07.030.
Heinze T., Koschella A. Polímeros, 2005, vol. 15(2), pp. 84–90. https://doi.org/10.1590/S0104-14282005000200005.
Amonova D.M. et al. Khimiya rastitel'nogo syr'ya, 2022, no. 2, pp. 51–60. https://doi.org/10.14258/jcprm.2022029551. (in Russ.).
Azimova L.B. et al. Russ. J. Bioorganic Chem., 2020, vol. 46, no. 7, 13. https://doi.org/10.1134/S106816202007002X.
Nunes C.A. et al. Food research international, 2015, vol. 75, pp. 270–280. https://doi.org/10.1016/j.foodres.2015.06.011.
Flores García Y. et al. Foods, 2025, vol. 14(9), 1587. https://doi.org/10.3390/foods14091587.
Tahmouzi S. et al. Food science & nutrition, 2023, vol. 11(9), pp. 4869–4897. https://doi.org/10.1002/fsn3.3383.
Gómez-Rodríguez G.N. et al. Biotecnia, 2023, vol. 25(2), pp. 126–135. https://doi.org/10.18633/biotecnia.v25i2.1883.
Shenoy M.A. et al. Journal of Applied Polymer Science, 2010, vol. 117(1), pp. 148–154. https://doi.org/10.1002/app.31872.
Wang J. et al. Carbohydrate polymers, 2014, vol. 113, pp. 325–335. https://doi.org/10.1016/j.carbpol.2014.07.028.
Cumpstey I. International scholarly research notices, 2013, vol. 1, 417672. https://doi.org/10.1155/2013/417672.
Heinze T. et al. Macromolecular Bioscience, 2016, vol. 16(1), pp. 10–42. https://doi.org/10.1002/mabi.201500184.
Copyright (c) 2026 Химия растительного сырья

Это произведение доступно по лицензии Creative Commons «Attribution» («Атрибуция») 4.0 Всемирная.

This work is licensed under a Creative Commons Attribution 4.0 International License.
Авторы, которые публикуются в данном журнале, соглашаются со следующими условиями:
1. Авторы сохраняют за собой авторские права на работу и передают журналу право первой публикации вместе с работой, одновременно лицензируя ее на условиях Creative Commons Attribution License, которая позволяет другим распространять данную работу с обязательным указанием авторства данной работы и ссылкой на оригинальную публикацию в этом журнале.
2. Авторы сохраняют право заключать отдельные, дополнительные контрактные соглашения на неэксклюзивное распространение версии работы, опубликованной этим журналом (например, разместить ее в университетском хранилище или опубликовать ее в книге), со ссылкой на оригинальную публикацию в этом журнале.
3. Авторам разрешается размещать их работу в сети Интернет (например, в университетском хранилище или на их персональном веб-сайте) до и во время процесса рассмотрения ее данным журналом, так как это может привести к продуктивному обсуждению, а также к большему количеству ссылок на данную опубликованную работу.















